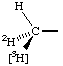
With the increasing use of isotopically substituted (i.e. 'carrier-free') compounds, especially in studies of the steric course of reactions, the conventions for specifying such compounds may now be of more importance to biochemists than they have been.
For isotopically substituted compounds the symbol for the isotope may be placed in a structural formula; it may also be placed in round brackets (parentheses) immediately before the name of the compounds, e.g.
(18O)ethanol, or CH3-CH2-18OH; similarly
(2-18O)glycolic acid, or H18O-CH2-COOH.
When such a compound is mixed with the corresponding compound of unmodified isotopic composition, the mixture is called an isotopically labelled compound. This can be indicated by specification of the isotope in square brackets before the name of the compound or within a formula, e.g.
[18O]ethanol, or CH3-CH2-[18O]H; similarly
[2-18O]glycolic acid, or H[18O]-CH2-COOH.
The two conventions may often need to be used together, e.g. for a chiral methyl group:
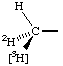
These rules make no changes in the use of square brackets long advocated in biochemical journals, but the practice with parentheses extends the scope of the system. Full details of the usage and extension to more complex situations are given in 'IUPAC Nomenclature of Organic Chemistry, Section H, Isotopically Modified Compounds' [Compendium, pp. 27-43; see also Eur. J. Biochem. 1978, 86, 9-25]. The definitive version has now been published [Pure Appl. Chem. 1979, 51, 353-380] and is also in the 1979 edition of Nomenclature of Organic Chemistry (Sections A, B, C, D, F, F, and H) published by Pergamon Press for IUPAC. It differs from the provisional version mainly in the addition of some extra possible components in the mixtures specified for multiply labelled compounds and in the addition of some clarifying statements [see Eur. J. Biochem. 1979, 102, 315-316].
The 'Compendium', mentioned above is the first edition of Biochemical nomenclature and related documents, The Biochemical Society, London 1978. This document was not reprinted in the second edition in 1992.
1. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN), and Nomenclature Commission of IUB (NC-IUB),.Newsletter 1980, Eur. J. Biochem., 1980, 104, 321-322; Hoppe-Seyler's Z. Physiol. Chem., 1980, 361, I-IV.
Return to the Newsletter home page